Double-jabbed’ Malhotra originally supported the program, until a series of events sent him digging into the evidence. What he discovered alarmed him and resulted in the publication of two evidence-based, peer-reviewed papers along with a call for the immediate suspension of the Covid mRNA roll-out…
Over following months, emerging data led Malhotra to question whether the vaccine was linked to his father’s death. The first was an abstract published in Circulation (November 8, 2021) by US cardiothoracic surgeon, Dr Steven Gundry, who followed several hundred of his patients after the mRNA (Moderna/Pfizer) jabs. Gundry found that inflammatory markers correlated with heart disease risk went through the roof. On average, that change increased the risk of those people having a heart attack or stroke within the next five years, from 11 per cent up to 25 per cent. This increase in risk is massive.
Efficacy
Browse the articles related to this topic below.
Join our community on Guilded.
Dr. John Campbell is an internet sensation, giving facts & data to help us navigate healthcare in the present day. The interview covers John’s development, work, rise to fame & what he would do to help get healthcare back in shape.
Moderna and the Food and Drug Association (FDA) have been accused of concealing data during the approval process for the pharma giant’s bivalent Covid booster.
Vaccine advisors who signed off on the updated shot late last year claim they were not shown trial data that indicated the booster was actually less effective at preventing Covid than the older vaccine it was meant to replace.
While the early trial results had substantial limitations, ‘disappointed’ and ‘angry advisors say its omission from panel discussions shows a remarkable lack of transparency.
The bivalent COVID-19 vaccine given to working-aged adults afforded modest protection overall against COVID-19, while the virus strains dominant in the community were those represented in the vaccine.
http://archive.today/2022.12.21-161132/https://www.medrxiv.org/content/10.1101/2022.12.17.22283625v1
RESULTS Of 1050 eligible HCW, 154 and 120 were enrolled to receive BNT162b2 and mRNA1273, respectively, and compared to 426 age-matched controls. Recipients of both vaccine types had a ∼9-10-fold increase in IgG and neutralizing titers within 2 weeks of vaccination and an 8-fold increase in live Omicron VOC neutralization, restoring titers to those measured after the third vaccine dose. Breakthrough infections were common, mostly very mild, yet, with high viral loads. Vaccine efficacy against infection was 30% (95%CI:-9% to 55%) and 11% (95%CI:-43% to +43%) for BNT162b2 and mRNA1273, respectively. Local and systemic adverse reactions were reported in 80% and 40%, respectively.
CONCLUSIONS The fourth COVID-19 mRNA dose restores antibody titers to peak post-third dose titers. Low efficacy in preventing mild or asymptomatic Omicron infections and the infectious potential of breakthrough cases raise the urgency of next generation vaccine development.
https://www.medrxiv.org/content/10.1101/2022.02.15.22270948v1.full-text
DOUBLE-JABBED Scots are now more likely to be admitted to hospital with Covid than the unvaccinated amid an increase in elderly people falling ill due to waning immunity.
It comes amid “weird” data showing that case rates have been lower in unvaccinated individuals than the single, double, or even triple-jabbed since Omicron became the dominant variant in Scotland.
The counterintuitive data from Public Health Scotland (PHS) contradicts previous pandemic trends which have consistently shown infection, hospitalisation and death rates to be highest among the unvaccinated.
Two doses of Pfizer vaccine offer limited protection, says Albert Bourla, Pfizer CEO.
Dr. Peter A. McCullough, MD, MPH, is a board-certified cardiologist who has testified before committees of the US and Texas Senate regarding the treatment of COVID-19 and management of the ongoing pandemic.
Backup mirrors:
Dec 10 (Reuters) – Most of the 43 COVID-19 cases caused by the Omicron variant identified in the United States so far were in people who were fully vaccinated, and a third of them had received a booster dose, according to a U.S. report published on Friday.
The U.S. Centers for Disease Control and Prevention (CDC) said that of the 43 cases attributed to Omicron variant, 34 people had been fully vaccinated. Fourteen of them had also received a booster, although five of those cases occurred less than 14 days after the additional shot before full protection kicks in.
While the numbers are very small, they add to growing concerns that current COVID-19 vaccines may offer less protection against the highly transmissible new variant.
Ten people have tested positive for Covid on board a Norwegian Cruise Line ship bound for New Orleans, despite the strict rules in place that required that all passengers and crew be vaccinated.
The vessel had departed the same city on November 28, and was scheduled to return there this weekend. On its route, the Norwegian Breakaway called at a number of ports in Belize, Honduras, and Mexico, according to the Louisiana Department of Health. More than 3,200 people are believed to be on board. Officials said in a statement that the cruise line “has been adhering to appropriate quarantine and isolation protocols as new cases and exposures have been identified.”
http://archive.today/2021.12.05-145243/https://www.rt.com/usa/542253-covid-outbreak-cruise-ship/
Data on confirmed Covid cases in England show how the Omicron variant is already taking hold in the country
Proportion of confirmed positive tests that didn’t detect the spike protein increased from 0.1% to 0.3%
One scientist said it equates around 60 more cases with hallmark than usual, suggesting Omicron infections
Comes as R rate spirals from less than one to 3.5 in South Africa’s Omicron epicentre Gauteng province
Lead UK epidemiologist says Omicron infections likely appear mild because of immunity from past infections
WHO officials suggested on Thursday that Covid cases were milder in those who caught the Omicron strain
However, despite this apparent evidence to support vaccine effectiveness – at least for the older age groups – on closer inspection of this data, this conclusion is cast into doubt. That is because we have shown a range of fundamental inconsistencies and flaws in the data. Specifically:
• In each group the non-Covid mortality rates in the three different categories of vaccinated people fluctuate in a wild, but consistent way, far removed from the expected historical mortality rates.
• Whereas the non-Covid mortality rate for unvaccinated should be consistent with historical mortality rates (and if, anything slightly lower than the vaccinated non-Covid mortality rate) it is not only higher than the vaccinated mortality rate, but it is far higher than the historical mortality rate.
• In previous years each of the 60-69, 70-79 and 80+ groups have mortality peaks at the same time during the year (including 2020 when all suffered the April Covid peak at the same time). Yet in 2021 each age group has non-Covid mortality peaks for the unvaccinated at a different time, namely the time that vaccination rollout programmes for those cohorts reach a peak.
• The peaks in the Covid mortality data for the unvaccinated are inconsistent with the actual Covid wave.
Whatever the explanations for the observed data, it is clear that it is both unreliable and misleading. We considered the socio-demographic and behavioural differences between vaccinated and unvaccinated that have been proposed as possible explanations for the data anomalies, but found no evidence supports any of these explanations. By Occam’s razor we believe the most likely explanations are:
• Systematic miscategorisation of deaths between the different groups of unvaccinated and vaccinated.
• Delayed or non-reporting of vaccinations.
• Systematic underestimation of the proportion of unvaccinated.
• Incorrect population selection for Covid deaths.
The analysis identified 72 studies that might potentially have provided evidence on the effectiveness of masks, social distancing and hand washing. Of those, just six (not eight, 30 or 72) were sufficiently relevant — and of sufficient quality — that they could provide any useful information on mask efficacy. And how reliable were the six? Four were assessed to have a moderate risk of bias, and two to have a serious or critical risk.
The boss of the drugmaker Moderna has warned that Covid-19 vaccines are unlikely to be as effective against the Omicron variant in comments that have added to uncertainty about its impact and unsettled financial markets.
“There is no world, I think, where [the effectiveness] is the same level we had with Delta,” Stéphane Bancel told the Financial Times. “I think it’s going to be a material drop. I just don’t know how much because we need to wait for the data. But all the scientists I’ve talked to … are like, ‘this is not going to be good’.”
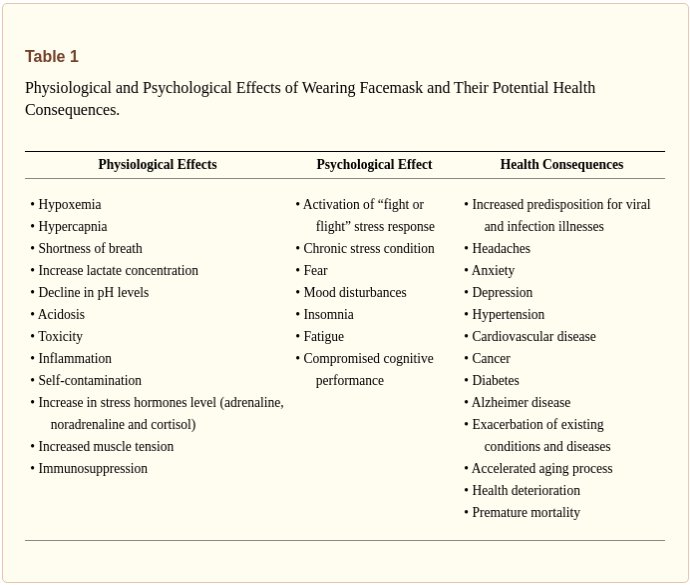
Each line in the above table describes “vaccine effectiveness”. That is how much the vaccine REDUCES chances of illness compared to the unvaccinated. The red numbers are NEGATIVE, meaning that vaccine INCREASES chances of geting Covid, compared to the unvaccinated. For example, for vaccinated 40-49 year olds, their chances of getting covid are (see above) 2.28 TIMES HIGHER than the unvaccinated. So their vaccine is a Covid magnet. No kidding.
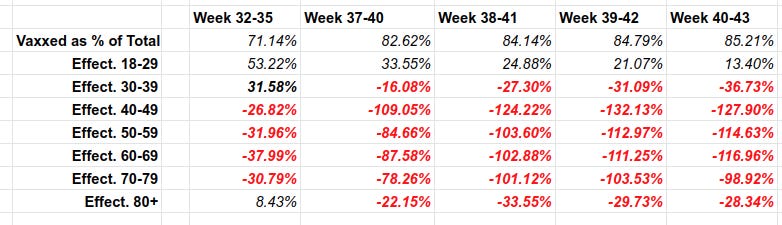
Over 82% of deaths in the UK are among the vaccinated.
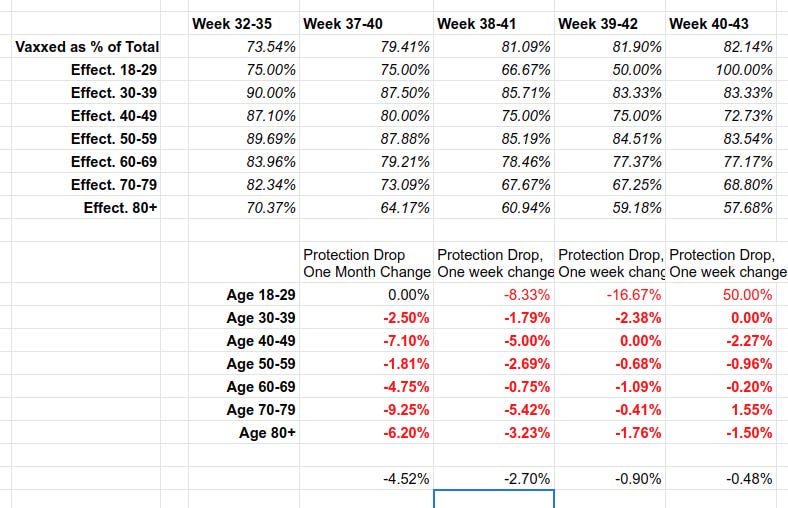
https://igorchudov.substack.com/p/uk-vaccines-weekly-ineffectiveness
To give you a preview of this article: Vaccines in the UK have become LESS THAN WORTHLESS in October. “The vaccinated” get infected MORE than the unvaccinated. The vaccinated have higher infection rates and are now the drivers of the UK epidemic. While some degree of “protection from death” still remains, it is dropping by about 7% every month for the most relevant groups. I break it down by age to avoid the stupid “but you need to do age adjustment” criticism, to show that the disaster is in almost every age group.
https://igorchudov.substack.com/p/uks-vaccine-hell-gets-worse-every
I reiterate our call: “slow down and get the science right—there is no legitimate reason to hurry to grant a license to a coronavirus vaccine.”
FDA should be demanding that the companies complete the two year follow-up, as originally planned (even without a placebo group, much can still be learned about safety). They should demand adequate, controlled studies using patient outcomes in the now substantial population of people who have recovered from covid. And regulators should bolster public trust by helping ensure that everyone can access the underlying data.
Many countries across the globe utilized medical and non-medical facemasks as non-pharmaceutical intervention for reducing the transmission and infectivity of coronavirus disease-2019 (COVID-19). Although, scientific evidence supporting facemasks’ efficacy is lacking, adverse physiological, psychological and health effects are established. Is has been hypothesized that facemasks have compromised safety and efficacy profile and should be avoided from use. The current article comprehensively summarizes scientific evidences with respect to wearing facemasks in the COVID-19 era, providing prosper information for public health and decisions making.
…The data suggest that both medical and non-medical facemasks are ineffective to block human-to-human transmission of viral and infectious disease such SARS-CoV-2 and COVID-19, supporting against the usage of facemasks. Wearing facemasks has been demonstrated to have substantial adverse physiological and psychological effects.