Despite the uncertainties, German Minister of Health Karl Lauterbach acknowledged in March that though rare, Long Covid–like symptoms after vaccination are a real phenomenon. He said his ministry was working to organize funding for studies, although none has been announced so far.
Researchers studying these complications also worry about undermining trust in COVID-19 vaccines. Harlan Krumholz, a cardiologist at Yale University, says concern that the antivaccine movement would seize on any research findings made him hesitant at first to dive in. But about a year ago he and Yale immunologist Akiko Iwasaki began to welcome postvaccination patients into a new study called LISTEN that also includes Long Covid patients. Among other things, it aims to correlate symptoms with immune cell patterns in blood samples.
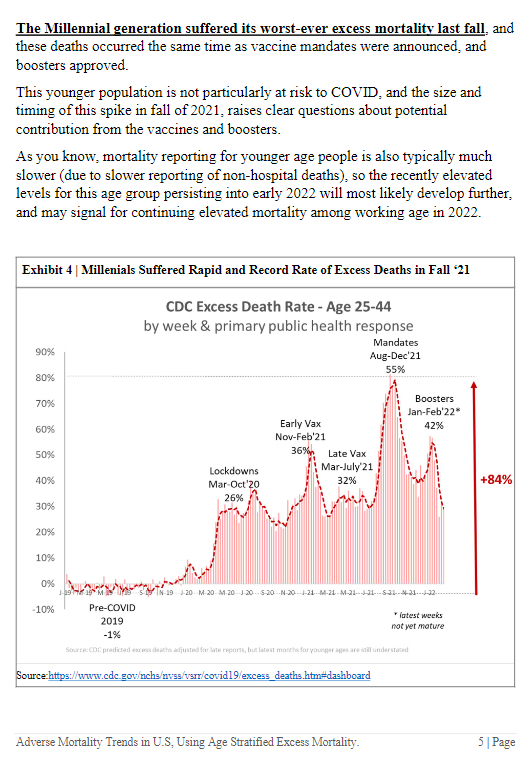
CDC
Browse the articles related to this topic below.
Join our community on Guilded.
- Misinformation #1: Natural immunity offers little protection compared to vaccinated immunity
- Misinformation #2: Masks prevent Covid transmission
- Misinformation #3: School closures reduce Covid transmission
- Misinformation #5: Young people benefit from a vaccine booster
- Misinformation #6: Vaccine mandates increased vaccination rates
- Misinformation #7: Covid originating from the Wuhan Lab is a conspiracy theory
- Misinformation #8: It was important to get the 2nd vaccine dose 3 or 4 weeks after the 1st dose
- Misinformation #8: It was important to get the 2nd vaccine dose 3 or 4 weeks after the 1st dose
- Misinformation #9: Data on the bivalent vaccine is “crystal clear”
- Misinformation #10: One in five people get long Covid
Published 20 Dec 2005.
We identified four fundamental questions underlying these claims and requested supporting studies from FLI (which according to the German Government “possesses virus isolates of H5N1”):
1.Does H5N1 exist?
2.Is it pathogenic to animals?
3.Is it transmissible and pathogenic to humans, and does it have pandemic potential?
4.Have other causes for observed disease been studied?
Our analysis shows the papers do not satisfy our four basic questions. Claims of H5N1 pathogenicity and pandemic potential need to be challenged further.
https://archive.today/2020.12.03-210342/https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7173052/
I was wrong. We in the scientific community were wrong. And it cost lives.
I can see now that the scientific community from the CDC to the WHO to the FDA and their representatives, repeatedly overstated the evidence and misled the public about its own views and policies, including on natural vs. artificial immunity, school closures and disease transmission, aerosol spread, mask mandates, and vaccine effectiveness and safety, especially among the young. All of these were scientific mistakes at the time, not in hindsight. Amazingly, some of these obfuscations continue to the present day.
Following the availability and use of the updated (bivalent) COVID-19 vaccines, CDC’s Vaccine Safety Datalink (VSD), a near real-time surveillance system, met the statistical criteria to prompt additional investigation into whether there was a safety concern for ischemic stroke in people ages 65 and older who received the Pfizer-BioNTech COVID-19 Vaccine, Bivalent. Rapid-response investigation of the signal in the VSD raised a question of whether people 65 and older who have received the Pfizer-BioNTech COVID-19 Vaccine, Bivalent were more likely to have an ischemic stroke in the 21 days following vaccination compared with days 22-44 following vaccination.
The Centers for Disease Control and Prevention (CDC) says that a preliminary COVID-19 vaccine “safety signal” has been identified and is investigating whether the Bivalent Pfizer-BioNTech vaccine creates an increased risk of ischemic stroke in people 65 and older.
In the Friday statement, the CDC said that the preliminary signal hasn’t been identified with the Bivalent Moderna COVID-19 vaccine.
Moderna and the Food and Drug Association (FDA) have been accused of concealing data during the approval process for the pharma giant’s bivalent Covid booster.
Vaccine advisors who signed off on the updated shot late last year claim they were not shown trial data that indicated the booster was actually less effective at preventing Covid than the older vaccine it was meant to replace.
While the early trial results had substantial limitations, ‘disappointed’ and ‘angry advisors say its omission from panel discussions shows a remarkable lack of transparency.
Academic freedom at Stanford is clearly dying. It cannot survive if the administration fails to create an environment where good-faith discussions can occur outside of a framework of ideological rigidity and the false certainties that ideologues—and governments—wish to impose on us. Stanford missed the opportunity to sponsor COVID policy forums and it deplatformed dissenting voices. Several prominent faculty exploited this environment, engaging in actions that directly violated basic academic norms.
Something strange is going on with the VAERS system. Reports that were present three months ago are now inexplicably missing. And fewer than 4% of adverse events recorded in V-Safe have made their way to VAERS. This is the CDC’s database; Dr. Rochelle Walensky is in charge of it. And the agency’s failure to properly manage VAERS is suppressing the already-alarming safety signal of the Covid-19 shots.
The vast majority of cases occur in young men, ages 16 to 24, according to the CDC. The agency did not have data available on the total number of cases in young adults 24 and younger, but it estimates there have been 52.4 cases and 56.3 cases per million doses of Pfizer’s and Moderna’s vaccines, respectively.
Symptoms of myocarditis include:
- Chest pain
- Shortness of breath
- Feelings of having a fast-beating, fluttering or pounding heart
A study by Canadian researchers published Monday in the Journal of the American College of Cardiology found that men younger than 40 who got the Moderna vaccine had the highest risk of heart issues, usually within 21 days after the second dose. The study was observational, meaning it doesn’t prove cause and effect but it is one of only a few studies to compare the risk of myocarditis between the Pfizer and the Moderna vaccines.
To help society mount a collective defence against pathogens, researchers say that leaders should enlist human-behaviour specialists to play a much bigger part in health policy. This has been the Achilles heel of governments during the COVID-19 pandemic, says Armand Balboni, an infectious-disease researcher and chief executive of pharmaceutical firm Appili Therapeutics in Halifax, Canada. “Social scientists, anthropologists and psychologists were not used nearly enough,” Balboni says.
http://archive.today/2022.11.15-082310/https://www.nature.com/articles/d41586-022-03354-8
On the contrary, over 30,000 Americans appear to have been killed by mechanical ventilators or other forms of medical iatrogenesis throughout April 2020, primarily in the area around New York.
This result is not altogether surprising, as subsequent studies revealed a 97.2% mortality rate among those over age 65 who were put on mechanical ventilators in accordance with the initial guidance from the WHO—as opposed to a 26.6% mortality rate among those over age 65 who weren’t put on mechanical ventilators—before a grassroots campaign put a stop to the practice by the beginning of May 2020.
As one doctor later told the Wall Street Journal, “We were intubating sick patients very early. Not for the patients’ benefit, but in order to control the epidemic… That felt awful.”
To put this in perspective, patients over age 65 were more than 26 times as likely to survive if they were not placed on mechanical ventilators.
https://michaelpsenger.substack.com/p/an-estimated-30000-americans-were
From 14 December 2020 through 31 May 2022 (persons 18–39 years) and 20 August 2022 (persons 5–17 years), 320 potential cases of myocarditis/pericarditis were identified 1 to 98 days after 6 992 340 vaccine doses as part of primary series COVID-19 vaccination, with 224 (70%) verified. Of these, 137 (61%) occurred 0 to 7 days after vaccination; 18 were after the first dose (of 3 562 311 doses administered) and 119 were after the second dose (of 3 430 029 doses administered).
In all age groups, incidence per million doses 0 to 7 days after vaccination was numerically higher in male than in female persons and after dose 2, although confidence intervals were wide and overlapped across sex for some age groups. Incidence was highest for male adolescents ages 12 to 15 years and 16 to 17 years following dose 2.
From 24 September 2021 through 20 August 2022, 101 potential cases of myocarditis/pericarditis were identified 1 to 98 days after 1 848 723 first booster doses, with 77 (76%) verified with a median onset of 4.5 days after vaccination; 39 cases (51%) were verified in the first week versus 38 during the subsequent 13 weeks.
In all age groups, incidence 0 to 7 days after first booster was higher for male compared to female persons, with adolescent males having the highest incidence in 16- to 17-year-olds and in 12- to 15-year-olds. In adults for whom both vaccine products were available, post-booster incidence was higher in male than in female adults and higher in males aged 18 to 29 compared to males aged 30 to 39.
http://archive.today/2022.10.06-094825/https://www.acpjournals.org/doi/10.7326/M22-2274
Thousands of American teenagers may have suffered heart inflammation after getting a Covid jab, a study suggests.
Researchers found up to one in 7,000 boys aged 12 to 15 years old developed myocarditis after receiving the Pfizer vaccine.
The condition — which is mild for most but can cause a recurrent heart palpitation in rare cases — was most common after the second dose.
http://archive.today/2022.10.03-220645/https://www.dailymail.co.uk/health/article-11275889/Up-one-7-000-American-teens-suffered-heart-inflammation-Covid-vaccine.html
…It also brings the recommendations for unvaccinated people in line with people who are fully vaccinated – an acknowledgment of the high levels of population immunity in the U.S., due to vaccination, past COVID-19 infections or both. “Based on the latest … data, it’s around 95% of the population,” Massetti said, “And so it really makes the most sense to not differentiate,” since many people have some protection against severe disease.
To prevent medically significant COVID-19 illness and death, persons must understand their risk, take steps to protect themselves and others with vaccines, therapeutics, and nonpharmaceutical interventions when needed, receive testing and wear masks when exposed, receive testing if symptomatic, and isolate for ≥5 days if infected.
http://archive.today/2022.08.12-074019/https://www.cdc.gov/mmwr/volumes/71/wr/mm7133e1.htm
FDA officials said in a statement that they decided to restrict Johnson & Johnson’s vaccine after taking another look at data on the risk of life-threatening blood clots within two weeks of vaccination.
Serco has won a £212m ($278m) contract for disease testing and contract tracing from the UK Health Security Agency, the organisations set up to replace the controversial NHS Test & Trace and doomed Public Health England.
In a contract initially set to last two years, the tech and public sector outsourcing provider will be expected to support services in the country including positive case tracing, contact tracing, isolation follow-up, test enquiries, and test bookings.
‘Former BlackRock Portfolio Manager, Investor Edward Dowd, Explains Bombshell ‘Fraud’ Charge re Pfizer Hiding Deaths Data; Many Media Outlets are ‘Accomplices to Murder’ DO NOT MISS. Historic.’
Edward Dowd Interview portion on Steve Bannons War Room Ep #1602
Two weeks ago the Centers for Disease Control and Prevention (CDC) published data about the effectiveness of boosters against COVID-19
The CDC failed to publish a tranche of their data, however – omitting the impact on those aged 18-49, who are least likely to benefit from boosters
The CDC are also being criticized for failing to publish their information about child hospitalization rates and comorbidities
A spokeswoman for the CDC said they were concerned that the data would be misinterpreted, pointing out that it was incomplete and not verified
Critics said that it was always better to publish the information rather than withhold, and allow scientists to analyze and explain what they could