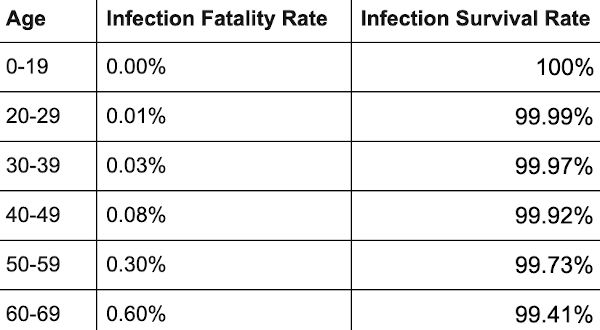
In August, Public Health England released data which shows that vaccination does not appreciably guard against Covid infection and transmission and protection worked out at around 17 per cent for the over-fifties. As I observed then, this would mean the vaxxed and unvaxxed pose a comparable danger to each other. All Covid apartheid schemes are therefore insensible.
Fresher information has fortified this conclusion of the summer. In every age group over 30 in the UK, the rates of Covid infection per 100,000 are now higher among the vaxxed than the unvaxxed. Indeed, in the cohorts aged between 40 and 79, infection rates among the vaccinated are more than twice as high as among the unvaccinated. PHE’s fruitlessly rechristened body, the UK Health Security Agency, frantically clarifies that the data ‘should not be used to estimate vaccine effectiveness’, a caveat which I include for the sake of accuracy. But the differences in the infection rates are drastic enough for you to draw your own conclusions.
Waning Immunity
Browse the articles related to this topic below.
Join our community on Guilded.
The elephants in the room – PANDA
While citizens focus on the latest issue the government directs our attention to, many of us have lost sight of the big picture and grown accustomed to severe limitations on our rights like the proverbial frogs in hot water. As a number of countries now seek to focus the discussion on further coercive measures and even mandatory vaccination, PANDA is increasingly aware of the elephants in the room, the topics no one is talking about.
http://archive.today/2021.09.16-104523/https://www.pandata.org/elephants-in-the-room/
I reiterate our call: “slow down and get the science right—there is no legitimate reason to hurry to grant a license to a coronavirus vaccine.”
FDA should be demanding that the companies complete the two year follow-up, as originally planned (even without a placebo group, much can still be learned about safety). They should demand adequate, controlled studies using patient outcomes in the now substantial population of people who have recovered from covid. And regulators should bolster public trust by helping ensure that everyone can access the underlying data.
