MPs have called for an urgent investigation into Britain’s soaring death rates as thousands more people than usual are dying each week.
Some 17,381 deaths were registered in England and Wales in the seven days to January 13 – 2,837 above average for the time of year.
This is the highest number of excess deaths since 3,429 in the week to February 12, 2021, when the UK was experiencing its second wave of Covid-19 infections and vaccination had only just begun.
On that occasion, deaths involving coronavirus accounted for 37 per cent of all those registered, according to the Office for National Statistics.
Pneumonia
Browse the articles related to this topic below.
Join our community on Guilded.
This guideline was developed before the COVID-19 pandemic. It covers diagnosing and managing pneumonia in adults who do not have COVID-19. It aims to improve accurate assessment and diagnosis of pneumonia to help guide antibiotic prescribing and ensure that people receive the right treatment.
July 2022: We reinstated this guideline, which was temporarily withdrawn in May 2020 because of the COVID-19 pandemic, and plan to update it.
https://web.archive.org/web/20220901083213/https://www.nice.org.uk/guidance/cg191
Dr. Philip McMillan interviews vaccine developer Geert Vanden Bossche and Robert Malone MD, inventor of mRNA vaccine platform.
Interview highlights
- Geert Vanden Bossche (GV) at 17m: Massive surges of the infection rates, especially in countries with an aggressive mass-vaccination policy, was predictable.
- Robert Malone (RM) at 23m: The Israeli data is a concern: we are seeing signs that the durability of the [Pfizer vaccine] is very poor.
- RM at 26m: The vaccinated are a higher risk of becoming superspreaders because they’re replicating virus at the same or higher levels than the unvaccinated but they feel better.
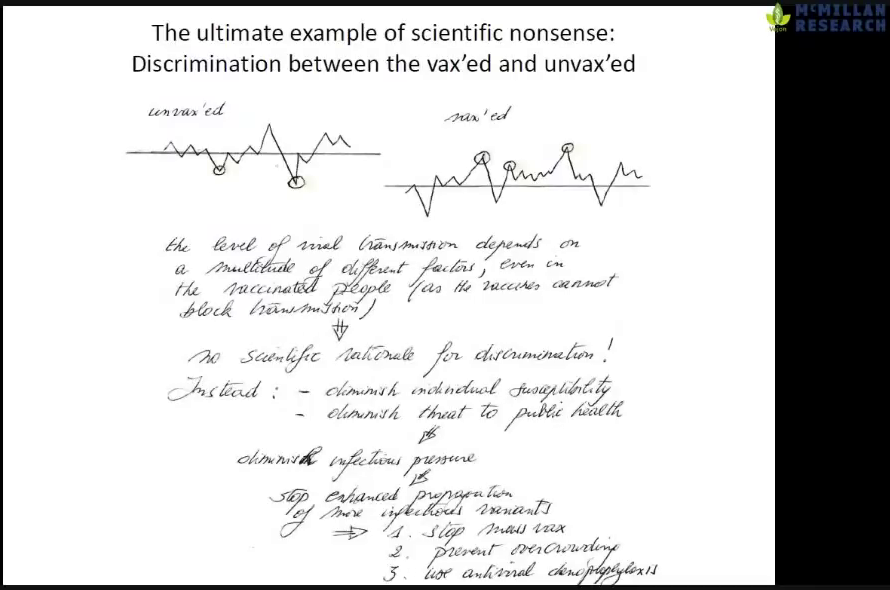
- GV at 28m: The effect of mass-vaccination is an ideal breeding-ground for more infections spread. However, if still have a substantial proportion that is non-vaccinated, you will see a reduction of infectious pressure.
- GV at 31m: The unvaccinated are ‘the vaccum cleaners’ who will eliminate a lot of virus from the population by mounting long-lived immunity and contribute to the reduction of infectious pressure. The vaccinated cannot contribute to the infectious pressure.
- RM at 33m: The truth is that it’s the vaccinated that are creating the risk, not the unvaccinated. The unvaccinated are serving as virus sinks. The probability of them having significant disease and death is minute. The real risk is the vaccinated who have received very focused spike proteins.
- GV at 35m: It is not a problem of individuals being vaccinated. The problem is a policy of mass-vaccination. That is how the more infections variant can adapt to the population and become dominant.
- GV at 58m: Young people are now getting the disease pretty fast because of the increased infectious pressure [due to mass-vaccination].
- RM at 1h10m: There are disincentives to asking questions about data for vaccine-enhanced replication and antibody-dependent enhancement; no-one wants fund the studies.
- GV at 1h12m: Regulators have no experience with the current situation where there are very many unknowns when deploying a new vaccine to the public.
- RM at 1h17m: The FDA is not structured to detect adverse advents and have admitted they cannot evaluate safety. Two of the top [US] regulators resigned because the FDA is no longer independent from the policy-making apparatus which exists in the Executive Branch [of US Government].
- RM at 1h24m: There is an intrinsic conflict of interest in the CDC in that it is funded to promote vaccines but also has the under-funded mission of evaluating their safety.
- RM at 1h25m: Policy recommendations together with Peter Navarro (American economist and author):
- Reserve vaccines for the high-risk population and make it available globally.
- Make early interventions [like Ivermectin and Vitamin D] widely available. Many are very effective when administered early and aggressively.
- Make home-test kits available (acknowledging that they have a bias to false positives) and make more specific tests in physicians offices.
- Address the fear by showing that currently most people are not at risk.
- GV at 1h30m: The most important thing is to reduce the infectious pressure. This is a huge threat to all those who were naturally protected, such as young people. The worst thing to do is to vaccinate the younger age groups because they are ‘the buffer’ of long-lived immunity. They are our hope for herd immunity. We will not get herd immunity from mass-vaccination.
- GV at 1h39m: We need to compare the ratio of severe disease of deaths in vaccinated and unvaccinated. We are seeing more case fatalities in the vaccinated but the numbers are not being made available.
- RM at 1h41m: There is a persistent signal in the UK data that there seems to be an excess deaths in the vaccinated and yet a relative deficit in the vaccinated. This is paradoxical.
- GV at 1h47m: Discrimination against the non-vaccinated is complete scientific nonsense. We should care about susceptibility. What is relevant is how can we protect ourselves best.
Slides:
Backup mirrors:
The kids are safe. They always have been.
It may sound strange, given a year of panic over school closures and reopenings, a year of masking toddlers and closing playgrounds and huddling in pandemic pods, that among children the mortality risk from COVID-19 is actually lower than from the flu. The risk of severe disease or hospitalization is about the same.
This is true for the much-worried-over Delta variant. It is also true for all the other variants, and for the original strain. Most remarkably, it has been known to be true since the very earliest days of the pandemic — indeed it was among the very first things we did know about the disease. The preliminary mortality data from China was very clear: To children, COVID-19 represented only a vanishingly tiny threat of death, hospitalization, or severe disease.
Yet for a year and a half we have been largely unwilling to fully believe it. Children now wear masks at little-league games, and at the swimming pool, and when school reopens in the fall they will likely wear masks there, too. But the kids are not at risk themselves, and never were. Now, thanks to vaccines, the vast majority of their parents and grandparents aren’t any longer, either.
https://nymag.com/intelligencer/2021/07/the-kids-were-safe-from-covid-the-whole-time.html
The number of people dying with flu and pneumonia on their death certificate in England and Wales is now 10 times higher than those with Covid, figures show.
The latest weekly data on deaths from the Office for National Statistics (ONS) show that there were 84 deaths mentioning Covid in the week ending June 11. There were 1,163 involving flu and pneumonia.
Registered Covid deaths fell by 14 per cent since the last update, in the week ending June 4, when 98 deaths were recorded.
Covid deaths now make up just 0.8 per cent of all deaths – down from 1.3 per cent in the previous week, despite the fact that week included the late May bank holiday, meaning there were fewer death registrations.
…The figures are much lower than would usually be expected for respiratory disease at this time of year. The five-year average for deaths involving flu and pneumonia in the same week is 1,704.
https://www.telegraph.co.uk/news/2021/06/22/flu-pneumonia-deaths-now-ten-times-higher-covid/
Medical and scientific experts now agree that bacteria, not influenza viruses, were the greatest cause of death during the 1918 flu pandemic.
…That pneumonia causes most deaths in an influenza outbreak is well known. Late 19th century physicians recognised pneumonia as the cause of death of most flu victims. While doctors limited fatalities in other 20th-century outbreaks with antibiotics such as penicillin, which was discovered in 1928, but did not see use in patients until 1942.
…McCullers’ research suggests that influenza kills cells in the respiratory tract, providing food and a home for invading bacteria. On top of this, an overstressed immune system makes it easier for the bacteria to get a foothold.
https://www.newscientist.com/article/dn14458-bacteria-were-the-real-killers-in-1918-flu-pandemic/
- Family said Captain Sir Tom Moore had regularly tested negative for Covid-19 until he visited hospital
- Only after a ten-day stay for pneumonia was he discharged on January 12 and then tested positive that day
- The inspirational NHS fundraiser later had to be readmitted and died in hospital surrounded by his family
- A family spokesman revealed Captain Tom’s pneumonia battle meant he had not had the coronavirus vaccine
- Centenarian became a national treasure over first coronavirus lockdown after raising millions for the NHS
- Boris Johnson today announced there would be a clap at 6pm by everyone to remember his achievements
- It came after a minute’s silence was held in the Commons by MPs in honour of the respected ex-army man
- His grandson Tom Teixeira also paid tribute to him, describing him as ‘a patriot to the country’
‘On January 22, Tom was discharged from hospital back to the family home where he felt most comfortable. Unfortunately he was left still fighting pneumonia and tested positive for Covid-19 that day.
‘Tom was able to have visitors to say goodbye to him at the end of his life. On Monday evening his daughter Hannah and grandchildren Benjie and Georgia were able to be by his side and his daughter Lucy was able to speak to him on FaceTime.’
Our mission: save the NHS by neglecting ourselves and the NHS. I received numerous CCG advice and flow-charts on the coronavirus-centric mass processing of patients. Most of it was about whom not to see, and who could pass the pearly gates of the hospitals. Then there was the advice on the parallel IT and video-consultation medical industrial revolution: our new NHS normal.
…For clarity, the “D” in coronavirus means “disease”, the second “S” in SARS-CoV-2 means “syndrome”. In a sense, the WHO had already decided Covid-19 was a distinct disease entity caused by a novel coronavirus before characterising it as a syndrome called SARS-2, and before the naming of the virus as SARS-CoV-2. The importance of scientific syntax and semantics cannot be overemphasised. Such cognitive slip-ups trickle unnoticed into general parlance and may have fatal consequences for us as a species.
Without a definite cause, one cannot definitively conclude to treat anything in particular. Is Covid-19 a syndrome, a mixed bag of symptoms and signs that has been negligently and politically globally fast-tracked to a scientifically wrong conclusion? Is it, in practice, a conflation of different, distinct disease entities including influenzae, rhinoviruses, pneumoniae and other coronaviruses, not to mention other non-infectious phenomena?
https://thecritic.co.uk/the-covid-physicians-true-coronavirus-timeline/
- AIDS was a testing pandemic, just like COVID-19.
- Many of the excess deaths for COVID-19 were due to inappropriately high dosages of hydroxychloroquine during experimental study trials.
- High COVID-19 excess deaths stopped after the trials were ended.
- Professor Martin Landry, leader of the UK-based Recovery trial, may have made a mistake in proposing high dosage of hydroxychloroquine. It seems he confused it with diiodohydroxyquinoline, treatment for treatment of amoebiasis.
- The treatment caused the damage.
- The danger of over-treatment is everywhere because the industry wants to sell diseases.
- COVID-19 is a self-limiting disease.
- The data shows that COVID-19 has no more killing potential than the yearly flu.
- Masks and lockdowns are ridiculous and damaging the whole population.
- It’s a political thing and not a health problem.
- Remdesivir is an immunosuppressant and useless against COVID-19.
- You have to live with viruses and you can’t fight against them.
- There is no treatment against COVID-19.
- The treatment against COVID-19 is to rest, like the flu.
- The problem is testing. If you stop the test, you’ll see nothing.
- Lockdowns were an overreaction.
- Vaccines are probably not a solution. You’ll have to vaccinate everyone every year. It’s good businesses.
There is no sign of a second coronavirus wave, experts have said as new Office for National Statistics (ONS) figures showed that deaths are just 1.5 per cent above the five-year average and tracking on a normal trajectory for the time of year.
Almost half of patients with COVID-19 have abnormal chest x-ray findings with peripheral GGO affecting the lower lobes being the most common finding. Chest x-ray can be used in diagnosis and follow up in patients with COVID-19 pneumonia.
https://bmcpulmmed.biomedcentral.com/articles/10.1186/s12890-020-01286-5
Nearly three times as many people are now dying of flu and pneumonia than with coronavirus in England and Wales, new figures have revealed.
Numbers published by the Office For National Statistics show 917 flu and pneumonia deaths were registered for the week ending on July 10.
In comparison, 366 people died that week after testing positive for Covid-19 – the lowest number of deaths involving the virus in the last 16 weeks and a 31.2% decrease compared with the previous week, which saw 532 deaths.
Overall, the number of deaths registered in the same week was 6.1% (560 deaths) below the five-year average – the fourth consecutive week it has been below average.
https://metro.co.uk/2020/07/22/nearly-three-times-people-dying-flu-pneumonia-coronavirus-13021417/
A normal chest radiograph does not exclude covid-19 pneumonia
No single feature of covid-19 pneumonia on a chest radiograph is specific or diagnostic, but a combination of multifocal peripheral lung changes of ground glass opacity and/or consolidation, which are most commonly bilateral, may be present
Diagnosis might be complicated as covid-19 pneumonia may or may not be visible on chest radiograph; consider other causes for patients’ respiratory symptoms
Note: This article, published on 16 October 2016, originally appeared in the Oral Health website. It was removed sometime after the end of June 2020 with no explanation other than, it being ‘no longer relevant in our current climate.’
The science behind face masks has not changed considerably in the past few months so we can only guess about what ‘no longer relevant’ means.
While you can find a capture at archive.org, we have saved a copy here to protect against censorship and for easy sharing.
Yesterday’s Scientific Dogma is Today’s Discarded Fable
Introduction
The above quotation is ascribed to Justice Archie Campbell author of Canada’s SARS Commission Final Report. 1 It is a stark reminder that scientific knowledge is constantly changing as new discoveries contradict established beliefs. For at least three decades a face mask has been deemed an essential component of the personal protective equipment worn by dental personnel. A current article, “Face Mask Performance: Are You Protected” gives the impression that masks are capable of providing an acceptable level of protection from airborne pathogens. 2 Studies of recent diseases such as Severe Acute Respiratory Syndrome (SARS), Middle Eastern Respiratory Syndrome (MERS) and the Ebola Crisis combined with those of seasonal influenza and drug resistant tuberculosis have promoted a better understanding of how respiratory diseases are transmitted. Concurrently, with this appreciation, there have been a number of clinical investigations into the efficacy of protective devices such as face masks. This article will describe how the findings of such studies lead to a rethinking of the benefits of wearing a mask during the practice of dentistry. It will begin by describing new concepts relating to infection control especially personal protective equipment (PPE).
Trends in Infection Control
For the past three decades there has been minimal opposition to what have become seemingly established and accepted infection control recommendations. In 2009, infection control specialist Dr. D. Diekema questioned the validity of these by asking what actual, front-line hospital-based infection control experiences were available to such authoritative organization as the Centers for Disease Control and Prevention (CDC), the Occupational Safety and Health Association (OSHA) and the National Institute for Occupational Safety and Health (NIOSH). 3 In the same year, while commenting on guidelines for face masks, Dr. M. Rupp of the Society for Healthcare Epidemiology of America noted that some of the practices relating to infection control that have been in place for decades, ”haven’t been subjected to the same strenuous investigation that, for instance, a new medicine might be subjected.” 4 He opined that perhaps it is the relative cheapness and apparent safety of face masks that has prevented them from undergoing the extensive studies that should be required for any quality improvement device. 4 More recently, Dr. R. MacIntyre, a prolific investigator of face masks, has forcefully stated that the historical reliance on theoretical assumptions for recommending PPEs should be replaced by rigorously acquired clinical data. 5 She noted that most studies on face masks have been based on laboratory simulated tests which quite simply have limited clinical applicability as they cannot account for such human factors as compliance, coughing and talking. 5
Covering the nose and mouth for infection control started in the early 1900s when the German physician Carl Flugge discovered that exhaled droplets could transmit tuberculosis. 4 The science regarding the aerosol transmission of infectious diseases has, for years, been based on what is now appreciated to be “very outmoded research and an overly simplistic interpretation of the data.” 6 Modern studies are employing sensitive instruments and interpretative techniques to better understand the size and distribution of potentially infectious aerosol particles. 6 Such knowledge is paramount to appreciating the limitations of face masks. Nevertheless, it is the historical understanding of droplet and airborne transmission that has driven the longstanding and continuing tradition of mask wearing among health professionals. In 2014, the nursing profession was implored to “stop using practice interventions that are based on tradition” but instead adopt protocols that are based on critical evaluations of the available evidence. 7
A December 2015 article in the National Post seems to ascribe to Dr. Gardam, Director of Infection Prevention and Control, Toronto University Health Network the quote, “I need to choose which stupid, arbitrary infection control rules I’m going to push.” 8 In a communication with the author, Dr. Gardam explained that this was not a personal belief but that it did reflect the views of some infection control practitioners. In her 2014 article, “Germs and the Pseudoscience of Quality Improvement”, Dr. K Sibert, an anaesthetist with an interest in infection control, is of the opinion that many infection control rules are indeed arbitrary, not justified by the available evidence or subjected to controlled follow-up studies, but are devised, often under pressure, to give the appearance of doing something. 9
The above illustrate the developing concerns that many infection control measures have been adopted with minimal supporting evidence. To address this fault, the authors of a 2007 New England Journal of Medicine (NEJM) article eloquently argue that all safety and quality improvement recommendations must be subjected to the same rigorous testing as would any new clinical intervention. 10 Dr. R. MacIntyre, a proponent of this trend in infection control, has used her research findings to boldly state that, “it would not seem justifiable to ask healthcare workers to wear surgical masks.” 4 To understand this conclusion it is necessary to appreciate the current concepts relating to airborne transmissions.
Airborne Transmissions
Early studies of airborne transmissions were hampered by the fact that the investigators were not able to detect small particles (less than 5 microns) near an infectious person. 6 Thus, they assumed that it was the exposure of the face, eyes and nose to large particles (greater than 5 microns) or “droplets” that transmitted the respiratory condition to a person in close proximity to the host. 6 This became known as “droplet infection”, and 5 microns or greater became established as the size of large particles and the traditional belief that such particles could, in theory, be trapped by a face mask. 5 The early researchers concluded that since only large particles were detected near an infectious person any small particles would be transmitted via air currents, dispersed over long distances, remain infective over time and might be inhaled by persons who never had any close contact with the host. 11 This became known as “airborne transmission” against which a face mask would be of little use. 5
Through the use of highly sensitive instruments it is now appreciated that the aerosols transmitted from the respiratory tract due to coughing, sneezing, talking, exhalation and certain medical and dental procedures produce respiratory particles that range from the very small (less than 5 microns) to the very large (greater than a 100 microns) and that all of these particles are capable of being inhaled by persons close to the source. 6, 11 This means that respiratory aerosols potentially contain bacteria averaging in size from 1-10 microns and viruses ranging in size from 0.004 to 0.1 microns. 12 It is also acknowledged that upon their emission large “droplets” will undergo evaporation producing a concentration of readily inhalable small particles surrounding the aerosol source. 6
The historical terms “droplet infection” and “airborne transmission” defined the routes of infection based on particle size. Current knowledge suggests that these are redundant descriptions since aerosols contain a wide distribution of particle sizes and that they ought to be replaced by the term, “aerosol transmissible.” 4, 5 Aerosol transmission has been defined as “person –to – person transmission of pathogens through air by means of inhalation of infectious particles.” 26 In addition, it is appreciated that the physics associated with the production of the aerosols imparts energy to microbial suspensions facilitating their inhalation. 11
Traditionally face masks have been recommended to protect the mouth and nose from the “droplet” route of infection, presumably because they will prevent the inhalation of relatively large particles. 11 Their efficacy must be re-examined in light of the fact that aerosols contain particles many times smaller than 5 microns. Prior to this examination, it is pertinent to review the defence mechanism of the respiratory tract.
Respiratory System Defences
Comprehensive details on the defence mechanisms of the respiratory tract will not be discussed. Instead readers are reminded that; coughing, sneezing, nasal hairs, respiratory tract cilia, mucous producing lining cells and the phagocytic activity of alveolar macrophages provide protection against inhaled foreign bodies including fungi, bacteria and viruses. 13 Indeed, the pathogen laden aerosols produced by everyday talking and eating would have the potential to cause significant disease if it were not for these effective respiratory tract defences.
These defences contradict the recently published belief that dentally produced aerosols, “enter unprotected bronchioles and alveoli.” 2 A pertinent demonstration of the respiratory tract’s ability to resist disease is the finding that- compared to controls- dentists had significantly elevated levels of antibodies to influenza A and B and the respiratory syncytial virus. 14 Thus, while dentists had greater than normal exposure to these aerosol transmissible pathogens, their potential to cause disease was resisted by respiratory immunologic responses. Interestingly, the wearing of masks and eye glasses did not lessen the production of antibodies, thus reducing their significance as personal protective barriers. 14 Another example of the effectiveness of respiratory defences is that although exposed to more aerosol transmissible pathogens than the general population, Tokyo dentists have a significantly lower risk of dying from pneumonia and bronchitis. 15 The ability of a face mask to prevent the infectious risk potentially inherent in sprays of blood and saliva reaching the wearers mouth and nose is questionable since, before the advent of mask use, dentists were no more likely to die of infectious diseases than the general population. 16
The respiratory tract has efficient defence mechanisms. Unless face masks have the ability to either enhance or lessen the need for such natural defences, their use as protection against airborne pathogens must be questioned.
Face Masks
History: Cloth or cotton gauze masks have been used since the late 19th century to protect sterile fields from spit and mucous generated by the wearer. 5,17,18 A secondary function was to protect the mouth and nose of the wearer from the sprays and splashes of blood and body fluids created during surgery. 17 As noted above, in the early 20th century masks were used to trap infectious “droplets” expelled by the wearer thus possibly reducing disease transmission to others. 18 Since the mid-20th century until to-day, face masks have been increasingly used for entirely the opposite function: that is to prevent the wearer from inhaling respiratory pathogens. 5,20,21 Indeed, most current dental infection control recommendations insist that a face mask be worn, “as a key component of personal protection against airborne pathogens”. 2
Literature reviews have confirmed that wearing a mask during surgery has no impact whatsoever on wound infection rates during clean surgery. 22,23,24,25,26 A recent 2014 report states categorically that no clinical trials have ever shown that wearing a mask prevents contamination of surgical sites. 26 With their original purpose being highly questionable it should be no surprise that the ability of face masks to act as respiratory protective devices is now the subject of intense scrutiny. 27 Appreciating the reasons for this, requires an understanding of the structure, fit and filtering capacity of face masks.
Structure and Fit: Disposable face masks usually consist of three to four layers of flat non-woven mats of fine fibres separated by one or two polypropylene barrier layers which act as filters capable of trapping material greater than 1 micron in diameter. 18,24,28 Masks are placed over the nose and mouth and secured by straps usually placed behind the head and neck. 21 No matter how well a mask conforms to the shape of a person’s face, it is not designed to create an air tight seal around the face. Masks will always fit fairly loosely with considerable gaps along the cheeks, around the bridge of the nose and along the bottom edge of the mask below the chin. 21 These gaps do not provide adequate protection as they permit the passage of air and aerosols when the wearer inhales. 11,17 It is important to appreciate that if masks contained filters capable of trapping viruses, the peripheral gaps around the masks would continue to permit the inhalation of unfiltered air and aerosols. 11
Filtering Capacity: The filters in masks do not act as sieves by trapping particles greater than a specific size while allowing smaller particles to pass through. 18 Instead the dynamics of aerosolized particles and their molecular attraction to filter fibres are such that at a certain range of sizes both large and small particles will penetrate through a face mask. 18 Accordingly, it should be no surprise that a study of eight brands of face masks found that they did not filter out 20-100% of particles varying in size from 0.1 to 4.0 microns. 21 Another investigation showed penetration ranges from 5-100% when masks were challenged with relatively large 1.0 micron particles. 29 A further study found that masks were incapable of filtering out 80-85% of particles varying in size from 0.3 to 2.0 microns. 30 A 2008 investigation identified the poor filtering performance of dental masks. 27 It should be concluded from these and similar studies that the filter material of face masks does not retain or filter out viruses or other submicron particles. 11,31 When this understanding is combined with the poor fit of masks, it is readily appreciated that neither the filter performance nor the facial fit characteristics of face masks qualify them as being devices which protect against respiratory infections. 27 Despite this determination the performance of masks against certain criteria has been used to justify their effectiveness.2 Accordingly, it is appropriate to review the limitations of these performance standards.
Performance Standards: Face masks are not subject to any regulations. 11 The USA Federal Food and Drug Administration (FDA) classifies face masks as Class II devices. To obtain the necessary approval to sell masks all that a manufacturer need do is satisfy the FDA that any new device is substantially the same as any mask currently available for sale. 21 As ironically noted by the Occupational Health and Safety Agency for Healthcare in BC, “There is no specific requirement to prove that the existing masks are effective and there is no standard test or set of data required supporting the assertion of equivalence. Nor does the FDA conduct or sponsor testing of surgical masks.” 21 Although the FDA recommends two filter efficiency tests; particulate filtration efficiency (PFE) and bacterial filtration efficiency (BFE) it does not stipulate a minimum level of filter performance for these tests. 27 The PFE test is a basis for comparing the efficiency of face masks when exposed to aerosol particle sizes between 0.1 and 5.0 microns. The test does not assess the effectiveness of a mask in preventing the ingress of potentially harmful particles nor can it be used to characterize the protective nature of a mask. 32 The BFE test is a measure of a mask’s ability to provide protection from large particles expelled by the wearer. It does not provide an assessment of a mask’s ability to protect the wearer. 17 Although these tests are conducted under the auspices of the American Society of Testing and Materials (ASTM) and often produce filtration efficiencies in the range of 95-98 %, they are not a measure of a masks ability to protect against respiratory pathogens. Failure to appreciate the limitations of these tests combined with a reliance on the high filtration efficiencies reported by the manufacturers has, according to Healthcare in BC, “created an environment in which health care workers think they are more protected than they actually are.” 21 For dental personnel the protection sought is mainly from treatment induced aerosols.
Dental Aerosols
For approximately 40 years it has been known that dental restorative and especially ultrasonic scaling procedures produce aerosols containing not only blood and saliva but potentially pathogenic organisms. 33 The source of these organisms could be the oral cavities of patients and/or dental unit water lines. 34 Assessing the source and pathogenicity of these organisms has proven elusive as it is extremely difficult to culture bacteria especially anaerobes and viruses from dental aerosols. 34 Although there is no substantiated proof that dental aerosols are an infection control risk, it is a reasonable assumption that if pathogenic microbes are present at the treatment site they will become aerosolized and prone to inhalation by the clinician which a face mask will not prevent. As shown by the study of UK dentists, the inhalation resulted in the formation of appropriate antibodies to respiratory pathogens without overt signs and symptoms of respiratory distress. 14 This occurred whether masks were or were not worn. In a 2008 article, Dr. S. Harrel, of the Baylor College of Dentistry, is of the opinion that because there is a lack of epidemiologically detectable disease from the use of ultrasonic scalers, dental aerosols appear to have a low potential for transmitting disease but should not be ignored as a risk for disease transmission. 34 The most effective measures for reducing disease transmission from dental aerosols are pre-procedural rinses with mouthwashes such as chlorhexidine, large diameter high volume evacuators, and rubber dam whenever possible. 33 Face masks are not useful for this purpose, and Dr. Harrel believes that dental personnel have placed too great a reliance on their efficacy. 34 Perhaps this has occurred because dental regulatory agencies have failed to appreciate the increasing evidence on face mask inadequacies.
The Inadequacies
Between 2004 and 2016 at least a dozen research or review articles have been published on the inadequacies of face masks. 5,6,11,17,19,20,21,25,26,27,28,31 All agree that the poor facial fit and limited filtration characteristics of face masks make them unable to prevent the wearer inhaling airborne particles. In their well-referenced 2011 article on respiratory protection for healthcare workers, Drs. Harriman and Brosseau conclude that, “facemasks will not protect against the inhalation of aerosols.” 11 Following their 2015 literature review, Dr. Zhou and colleagues stated, “There is a lack of substantiated evidence to support claims that facemasks protect either patient or surgeon from infectious contamination.” 25 In the same year Dr. R. MacIntyre noted that randomized controlled trials of facemasks failed to prove their efficacy. 5 In August 2016 responding to a question on the protection from facemasks the Canadian Centre for Occupational Health and Safety replied:
- The filter material of surgical masks does not retain or filter out submicron particles;
- Surgical masks are not designed to eliminate air leakage around the edges;
- Surgical masks do not protect the wearer from inhaling small particles that can remain airborne for long periods of time. 31
In 2015, Dr. Leonie Walker, Principal Researcher of the New Zealand Nurses Organization succinctly described- within a historical context – the inadequacies of facemasks, “Health care workers have long relied heavily on surgical masks to provide protection against influenza and other infections. Yet there are no convincing scientific data that support the effectiveness of masks for respiratory protection. The masks we use are not designed for such purposes, and when tested, they have proved to vary widely in filtration capability, allowing penetration of aerosol particles ranging from four to 90%.” 35
Face masks do not satisfy the criteria for effectiveness as described by Drs. Landefeld and Shojania in their NEJM article, “The Tension between Needing to Improve Care and Knowing How to Do It. 10 The authors declare that, “…recommending or mandating the widespread adoption of interventions to improve quality or safety requires rigorous testing to determine whether, how, and where the intervention is effective…” They stress the critical nature of this concept because, “…a number of widely promulgated interventions are likely to be wholly ineffective, even if they do not harm patients.” 10 A significant inadequacy of face masks is that they were mandated as an intervention based on an assumption rather than on appropriate testing.
Conclusions
The primary reason for mandating the wearing of face masks is to protect dental personnel from airborne pathogens. This review has established that face masks are incapable of providing such a level of protection. Unless the Centers for Disease Control and Prevention, national and provincial dental associations and regulatory agencies publically admit this fact, they will be guilty of perpetuating a myth which will be a disservice to the dental profession and its patients. It would be beneficial if, as a consequence of the review, all present infection control recommendations were subjected to the same rigorous testing as any new clinical intervention. Professional associations and governing bodies must ensure the clinical efficacy of quality improvement procedures prior to them being mandated. It is heartening to know that such a trend is gaining a momentum which might reveal the inadequacies of other long held dental infection control assumptions. Surely, the hallmark of a mature profession is one which permits new evidence to trump established beliefs. In 1910, Dr. C. Chapin, a public health pioneer, summarized this idea by stating, “We should not be ashamed to change our methods; rather, we should be ashamed not to do so.” 36 Until this occurs, as this review has revealed, dentists have nothing to fear by unmasking. OH
Oral Health welcomes this original article.
References
1. Ontario Ministry of Health and Long-term Care. SARS Commission-Spring of Fear: Final Report. Available at: http://www.health.gov.on.ca/english/public/pub/ministry_reports/campbell06/campbell06.html
2. Molinari JA, Nelson P. Face Mask Performance: Are You Protected? Oral Health, March 2016.
3. Diekema D. Controversies in Hospital Infection Prevention, October, 2009.
4. Unmasking the Surgical Mask: Does It Really Work? Medpage Today, Infectious Disease, October, 2009.
5. MacIntyre CR, Chughtai AA. Facemasks for the prevention of infection in healthcare and community settings. BMJ 2015; 350:h694.
6. Brosseau LM, Jones R. Commentary: Health workers need optimal respiratory protection for Ebola. Center for Infectious Disease Research and Policy. September, 2014.
7. Clinical Habits Die Hard: Nursing Traditions Often Trump Evidence-Based Practice. Infection Control Today, April, 2014.
8. Landman K. Doctors, take off those dirty white coats. National Post, December 7, 2015.
9. Sibert K. Germs and the Pseudoscience of Quality Improvement. California Society of Anesthesiologists, December 8, 2014.
10. Auerbach AD, Landfeld CS, Shojania KG. The Tension between Needing to Improve Care and Knowing How to Do It. NEJM 2007; 357 (6):608-613.
11. Harriman KH, Brosseau LM. Controversy: Respiratory Protection for Healthcare Workers. April, 2011. Available at: http://www.medscape.com/viewarticle/741245_print
12. Bacteria and Viruses Issues. Water Quality Association, 2016. Available at: https://www.wqa.org/Learn-About-Water/Common-Contaminants/Bacteria-Viruses
13. Lechtzin N. Defense Mechanisms of the Respiratory System. Merck Manuals, Kenilworth, USA, 2016
14. Davies KJ, Herbert AM, Westmoreland D. Bagg J. Seroepidemiological study of respiratory virus infections among dental surgeons. Br Dent J. 1994; 176(7):262-265.
15. Shimpo H, Yokoyama E, Tsurumaki K. Causes of death and life expectancies among dentists. Int Dent J 1998; 48(6):563-570.
16. Bureau of Economic Research and Statistics, Mortality of Dentists 1961-1966. JADA 1968; 76(4):831-834.
17. Respirators and Surgical Masks: A Comparison. 3 M Occupational Health and Environment Safety Division. Oct. 2009.
18. Brosseau L. N95 Respirators and Surgical Masks. Centers for Disease Control and Prevention. Oct. 2009.
19. Johnson DF, Druce JD, Birch C, Grayson ML. A Quantitative Assessment of the Efficacy of Surgical and N95 Masks to Filter Influenza Virus in Patients with Acute Influenza Infection. Clin Infect Dis 2009; 49:275-277.
20. Weber A, Willeke K, Marchloni R et al. Aerosol penetration and leakage characteristics of masks used in the health care industry. Am J Inf Cont 1993; 219(4):167-173.
21. Yassi A, Bryce E. Protecting the Faces of Health Care Workers. Occupational Health and Safety Agency for Healthcare in BC, Final Report, April 2004.
22. Bahli ZM. Does Evidence Based Medicine Support The Effectiveness Of Surgical Facemasks In Preventing Postoperative Wound Infections In Elective Surgery. J Ayub Med Coll Abbottabad 2009; 21(2)166-169.
23. Lipp A, Edwards P. Disposable surgical face masks for preventing surgical wound infection in clean surgery. Cochrane Database Syst Rev 2002(1) CD002929.
24. Lipp A, Edwards P. Disposable surgical face masks: a systematic review. Can Oper Room Nurs J 2005; 23(#):20-38.
25. Zhou Cd, Sivathondan P, Handa A. Unmasking the surgeons: the evidence base behind the use of facemasks in surgery. JR Soc Med 2015; 108(6):223-228.
26. Brosseau L, Jones R. Commentary: Protecting health workers from airborne MERS-CoV- learning from SARS. Center for Infectious Disease Research and Policy May 2014.
27. Oberg T, Brosseau L. Surgical mask filter and fit performance. Am J Infect Control 2008; 36:276-282.
28. Lipp A. The effectiveness of surgical face masks: what the literature shows. Nursing Times 2003; 99(39):22-30.
29. Chen CC, Lehtimaki M, Willeke K. Aerosol penetration through filtering facepieces and respirator cartridges. Am Indus Hyg Assoc J 1992; 53(9):566-574.
30. Chen CC, Willeke K. Characteristics of Face Seal Leakage in Filtering Facepieces. Am Indus Hyg Assoc J 1992; 53(9):533-539.
31. Do surgical masks protect workers? OSH Answers Fact Sheets. Canadian Centre for Occupational health and Safety. Updated August 2016.
32. Standard Test Method for Determining the Initial Efficiency of Materials Used in Medical Face Masks to Penetration by Particulates Using Latex Spheres. American Society of Testing and Materials, Active Standard ASTM F2299/F2299M.
33. Harrel SK. Airborne Spread of Disease-The Implications for Dentistry. CDA J 2004; 32(11); 901-906.
34. Harrel SK. Are Ultrasonic Aerosols an Infection Control Risk? Dimensions of Dental Hygiene 2008; 6(6):20-26.
35. Robinson L. Unmasking the evidence. New Zealand Nurses Organization. May 2015. Available at: https://nznoblog.org.nz/2015/05/15/unmasking-the-evidence
36. Chapin CV. The Sources and Modes of Transmission. New York, NY: John Wiley & Sons; 1910.