It is very hard to tell the difference between Covid and a cold without testing
Before the Covid-19 pandemic, if you got a sniffle and a headache, you might dismiss it as an ordinary cold and carry on as normal, even if you felt a little rough around the edges. But during cold and flu season, how can you be sure it’s a cold and not the coronavirus?
The common cold is caused by a different strain of virus to Covid-19. But with the rapidly-spreading Omicron variant often causing mild symptoms, such as stuffy nose, sore head and sore throat, it is very hard to tell the difference without testing.
Independent
Browse the articles related to this topic below.
Join our community on Guilded.
Scotland international Siobhan Cattigan has died aged 26, Scottish Rugby has announced.
Funeral firm Dignity said profits have fallen amid a marked drop in UK deaths since April as the group’s new boss outlined his turnaround plans.
The group said that following a 27% surge in first quarter deaths to 204,000 during a Covid-hit start to the year, the number of deaths fell below the five-year average in April and May and were 7% lower.
This has left underlying operating profits “slightly” lower year-on-year at £30.7 million for the 21 weeks to May 21, according to Dignity.
Hospitals have been told to change the way they collect data on patients infected with coronavirus to differentiate between those actually sick with symptoms and those who test positive while seeking treatment for something else.
The move would reduce the overall number of patients in hospital for coronavirus as until now data from hospitals has included all patients who tested positive for Covid-19, regardless of whether they had symptoms or not.
https://www.independent.co.uk/news/health/coronavirus-hospitals-nhs-england-data-b1862804.html
But the time has come when the hard choices are looming closer. If we don’t want this Covid crisis to last forever, we need some new simple, guidelines: No jab, no job; no jab, no access to NHS healthcare; no jab, no state education for your kids. No jab, no access to pubs, restaurants, theatres, cinemas, stadiums. No jab, no entry to the UK, and much else.
https://www.independent.co.uk/voices/antivaxxers-vaccine-coronavirus-nhs-b1849437.html
Germany has recommended the AstraZeneca coronavirus vaccine should only be given to people under the age of 65, according to reports.
The country’s vaccine committee has reportedly said the jab, developed alongside the University of Oxford, should only be offered to people aged between 18 and 64.
It gave a lack of sufficient data on the effectiveness of the inoculation in older people as the reason for the decision in a draft recommendation.
Hospitals have spent £2 million on more than 50 gagging orders preventing staff speaking out, a Freedom of Information Act request has revealed.
Tory MP Steve Barclay, who obtained the figures, accused NHS chief Sir David Nicholson of either failing to ask questions about the orders or being “complicit in a cover-up”.
Sir David will retire as NHS England’s chief executive next year but Mr Barclay said he should stand down now because the culture in the health service had to change.
It was reported last night that at least 52 staff have been silenced using the orders since 2008, some of which cost as much as £500,000. All are thought to contain confidentiality clauses.
…North East Cambridgeshire MP Mr Barclay told The Daily Telegraph: “It is simply not plausible that the man who was supposed to be running the NHS was seemingly unaware that employees threatening to speak out were being offered golden goodbyes in return for a vow of silence.”
Secret talks covered vaccines and mass testing
Tony Blair has been advising health secretary Matt Hancock on coronavirus pandemic strategy, it has emerged.
The former Labour prime minister has reportedly privately offered strategic advice to the government a number of times during the course of the outbreak.
Topics touched on by the ex-Labour leader are said to include vaccine strategy and mass testing, the Sunday Times newspaper reported.
The secret talks are reportedly part of an attempted comeback for the former politician, according to a source familiar with his thinking who spoke to the same newspaper.
The UK government has granted pharmaceutical giant Pfizer a legal indemnity protecting it from being sued, enabling its coronavirus vaccine to be rolled out across the country as early as next week.
The Department of Health and Social Care has confirmed the company has been given an indemnity protecting it from legal action as a result of any problems with the vaccine.
Ministers have also changed the law in recent weeks to give new protections to companies such as Pfizer, giving them immunity from being sued by patients in the event of any complications.
NHS staff providing the vaccine, as well as manufacturers of the drug, are also protected.
No traces of coronavirus have been found on surfaces and in the air on the London Underground or on buses in the capital city, scientists have said.
Experts from Imperial College London have been carrying out monthly tests on the network, mimicking a passenger journey and taking swabs from escalators, handrails, bus shelters and Oyster Card readers.
Article date: Monday 6 April 2009
At stake at one point last year was more than $8bn in punitive damages being sought in a string of cases, as well as potential jail terms in Nigeria for several Pfizer staff. “There has been a complex web of cases with proceedings in Connecticut, New York, Lagos, Abuja and Kano,” Mr Etigwe said. “The strategy of big companies when they are dealing with smaller opponents is to stretch the process, to overwhelm us until we are ready to accept whatever they want to offer.” Trovan never became the blockbuster that Pfizer had hoped for and it is no longer in production. The EU has banned the drug and it has been withdrawn from sale in the US.
Almost 60 per cent of staff infected with coronavirus continued to work and commute
Clinical trials:
- Phase I clinical trials simply test the safety of a drug or vaccine in a small number of healthy volunteers — usually brave and naïve college students.
- Phase II trials are responsible for testing its effectiveness in a larger number of subjects.
- A hyped-up and exuberant response to a Phase I trial as seen with Moderna press release is rare and nearly unheard of.
- Little information is gleaned from an investigational drug in Phase I that has many more hurdles to overcome before it successfully gets to market
- 77 percent of vaccines for infectious diseases make it through Phase I, but only 33 percent make it through the entire process overall.
Moderna’s RNA vaccine
- Upon examining Moderna’s non-peer reviewed press release, the actual data on the vaccine’s success is even more flimsy.
- When it comes to finding out whether the vaccine elicits an antibody response that could potentially fight the coronavirus, they only had data on eight patients out of the 45 patients who received the vaccine.
- The only data Moderna mentioned when it comes to determining whether the vaccine was clinically effective against the coronavirus were from mice.
- History also proves that success in animal models is often not replicated in human studies.
- Moderna’s messenger RNA vaccine is completely new and revolutionary. Messenger RNA vaccines have never before been brought to market for human patients
- It uses a sequence of genetic RNA material produced in a lab that, when injected into your body, must invade your cells and hijack your cells’ protein-making machinery called ribosomes to produce the viral components that subsequently train your immune system to fight the virus.
- Some messenger RNA vaccines are self-amplifying. That means they can force the cell to replicate more copies of itself.
- There are unique and unknown risks to messenger RNA vaccines, including the possibility that they generate strong type I interferon responses that could lead to inflammation and autoimmune conditions.
Oxford Vaccine Group’s vaccine:
- Oxford Vaccine Group has a competing vaccine that does not need to invade and hijack our cells’ own machinery.
- From a medical and clinical perspective, there is less risk of generating a type I interferon response and autoimmunity because there is no messenger RNA floating around our blood, invading our cells.
Members of the public could be putting themselves more at risk from contracting coronavirus by wearing face masks, one of England’s most senior doctors has warned.
Jenny Harries, deputy chief medical officer, said the masks could “actually trap the virus” and cause the person wearing it to breathe it in.
“For the average member of the public walking down a street, it is not a good idea” to wear a face mask in the hope of preventing infection, she added.
Jake Dunning, head of emerging infections and zoonoses [infectious disease spread between humans and animals] at Public Health England, told The Independent there was “very little evidence of a widespread benefit” from wearing them.
During the Cold War, the British Government used the general public as unwitting biological and chemical warfare guinea pigs on a much greater scale than previously thought, according to new historical research.
In more than 750 secret operations, hundreds of thousands of ordinary Britons were subjected to ‘mock’ biological and chemical warfare attacks launched from aircraft, ships and road vehicles.
Up until now historians had thought that such operations had been much less extensive. The new research, carried out by Ulf Schmidt, Professor of Modern History at the University of Kent, has revealed that British military aircraft dropped thousands of kilos of a chemical of ‘largely unknown toxic potential’ on British civilian populations in and around Salisbury in Wiltshire, Cardington in Bedfordshire and Norwich in Norfolk.
It was the worst winter on record for more than 40 years, with the 1975-76 season being the last time deaths climbed so high above the expected levels.
The NHS was rocked by a record winter crisis in early 2018, with a massive rise in flu cases and sub-zero temperatures triggered by the Beast from the East storm, which added further to death rates.
“The number of excess winter deaths in England and Wales in 2017 to 2018 was the highest recorded since the winter of 1975 to 1976,” said Nick Stripe, from the ONS Health Analysis and Life Events team.”
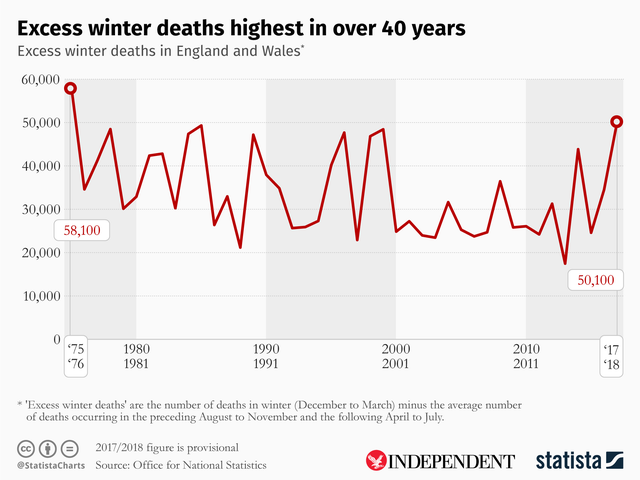
The flu vaccine’s failure to protect against some of the key strains of the infection contributed to more than 50,000 “extra” deaths in England and Wales last winter, according to data from the Office of National Statistics.