Austria’s powerful Constitutional Court has demanded detailed data from the country’s Health Ministry justifying the government’s coronavirus response.
The 14-member court issued 10 sets of questions to the Health Ministry on January 26 in order to prepare for a “possible oral hearing” into a number of complaints it has received against Austria’s Covid-19 measures.
Court Cases
Browse the articles related to this topic below.
Join our community on Guilded.
The Associated Press recently ran a story they said debunked the dissenting Covid concerns of pathologist, Dr. Roger Hodkinson. In their article titled, “Pathologist falsely claims COVID-19 is a hoax, no worse than the flu,” they misrepresented several of Dr. Hodkinson’s statements. The also wrote specifically saying they were planning to debunk him, not understand what he meant. Dr Hodkinson is a medical specialist in pathology and graduate of Cambridge University, UK. He is a Fellow of the College of American Pathologists and the Royal College of Physicians and Surgeons of Canada. He was previously the President of the Alberta Society of Laboratory Physicians, an Assistant Professor in the Faculty of Medicine at the University of Alberta, and CEO of a large community based medical laboratory with a full menu of testing for infectious disease and virology. He is currently the Chairman of an American biotechnology company active in DNA sequencing.
Dr Hodkinson is the CEO of Western Medical Assessments, and has been the Company’s Medical Director for over 20 years. He received his general medical degrees from Cambridge University in the UK, and then became a Royal College certified pathologist in Canada (FRCPC) following a residency in Vancouver, BC.
Source: Western Medical Assessments
- Dr Hodkinson’s interview is at 2h49m.
Find out more about the Investigative Corona Committee Germany.

Backup mirrors:
A shortened version of the video Session 56 has been uploaded to Bitchute by Coronavirus Plushie.
Dr. Michael Dykta, Dr. Wolfgang Wodarg, Professor Arne Burkhardt, Dr. Peter McCullough and Dr. Roger Hodkinson speak with Dr. Reiner Fuellmich on Germany’s Investigative Corona Committee Session 56.
- Dr McCullough’s is at around 1h35m.
- Dr Hodkinson’s interview is at 2h49m.
Find out more about the Investigative Corona Committee Germany.
The full video has been removed from YouTube. You can find a backup mirror below:
Published 19 August 1992
New York-based Pfizer said the agreement will cost it between $165 million and $215 million, on a pretax basis, and will be offset by proceeds from the sale of most of its Shiley Inc. assets earlier this year as well as expected insurance reimbursements.
Published 9 September 2009
WASHINGTON – American pharmaceutical giant Pfizer Inc. and its subsidiary Pharmacia & Upjohn Company Inc. (hereinafter together “Pfizer”) have agreed to pay $2.3 billion, the largest health care fraud settlement in the history of the Department of Justice, to resolve criminal and civil liability arising from the illegal promotion of certain pharmaceutical products, the Justice Department announced today.
Anger at deadly Nigerian drug trials – BBC
Published 20 June 2007
Pfizer tested the then unregistered drug in Nigeria’s north-western Kano State during an outbreak of meningitis which had affected thousands of children.
Officials in Kano say more than 50 children died in the experiment, while many others developed mental and physical deformities.
…After more than a decade of silence, the Nigerian government has decided to sue Pfizer, seeking $7bn (£3.5bn) in damages for the families of children who allegedly died or suffered side-effects in the experiment.
Wikipedia snapshot from 9 March 2021:
| Year | Company | Settlement | Violation(s) | Product(s) | Laws allegedly violated (if applicable) |
|---|---|---|---|---|---|
| 2012 | GlaxoSmithKline[1][6] | $3 billion ($1B criminal, $2B civil) | Criminal: Off-label promotion, failure to disclose safety data. Civil: paying kickbacks to physicians, making false and misleading statements concerning the safety of Avandia, reporting false best prices and underpaying rebates owed under the Medicaid Drug Rebate Program | Avandia (not providing safety data), Wellbutrin, Paxil (promotion of paediatric use), Advair, Lamictal, Zofran, Imitrex, Lotronex, Flovent, Valtrex | False Claims Act, FDCA |
| 2009 | Pfizer[2] | $2.3 billion | Off-label promotion, kickbacks | Bextra, Geodon, Zyvox, Lyrica | False Claims Act, FDCA |
| 2013 | Johnson & Johnson[7] | $2.2 billion | Off-label promotion, kickbacks | Risperdal, Invega, Nesiritide | False Claims Act, FDCA |
| 2012 | Abbott Laboratories[8] | $1.5 billion | Off-label promotion | Depakote | False Claims Act, FDCA |
| 2009 | Eli Lilly[9] | $1.4 billion | Off-label promotion | Zyprexa | False Claims Act, FDCA |
| 2001 | TAP Pharmaceutical Products[10] | $875 million | Medicare fraud, kickbacks | Lupron | False Claims Act, Prescription Drug Marketing Act |
| 2012 | Amgen[11] | $762 million | Off-label promotion, kickbacks | Aranesp | False Claims Act, FDCA |
| 2010 | GlaxoSmithKline[12] | $750 million | Poor manufacturing practices | Kytril, Bactroban, Paxil CR, Avandamet | False Claims Act, FDCA |
| 2005 | Serono[13] | $704 million | Off-label promotion, kickbacks, monopolistic practices | Serostim | False Claims Act |
| 2008 | Merck[14] | $650 million | Medicare fraud, kickbacks | Zocor, Vioxx, Pepsid | False Claims Act, Medicaid Rebate Statute |
| 2007 | Purdue Pharma[15] | $601 million | Off-label promotion | Oxycontin | False Claims Act |
| 2010 | Allergan[16] | $600 million | Off-label promotion | Botox | False Claims Act, FDCA |
| 2010 | AstraZeneca[17] | $520 million | Off-label promotion, kickbacks | Seroquel | False Claims Act |
| 2007 | Bristol-Myers Squibb[18] | $515 million | Off-label promotion, kickbacks, Medicare fraud | Abilify, Serzone | False Claims Act, FDCA |
| 2002 | Schering-Plough[19] | $500 million | Poor manufacturing practices | Claritin | FDA Current Good Manufacturing Practices |
| 2006 | Mylan[20] | $465 million | Misclassification under the Medicaid Drug Rebate Program | EpiPen (epinephrine) | False Claims Act |
| 2006 | Schering-Plough[21] | $435 million | Off-label promotion, kickbacks, Medicare fraud | Temodar, Intron A, K-Dur, Claritin RediTabs | False Claims Act, FDCA |
| 2004[22] | Pfizer | $430 million | Off-label promotion | Neurontin | False Claims Act, FDCA |
| 2008 | Cephalon[23] | $425 million | Off-label promotion[23] | Actiq, Gabitril, Provigil | False Claims Act, FDCA |
| 2010 | Novartis[24] | $423 million | Off-label promotion, kickbacks | Trileptal | False Claims Act, FDCA |
| 2003 | AstraZeneca[25] | $355 million | Medicare fraud | Zoladex | Prescription Drug Marketing Act |
| 2004 | Schering-Plough[26] | $345 million | Medicare fraud, kickbacks | Claritin | False Claims Act, Anti-Kickback Statute |
Last September, Pfizer’s CEO, Albert Bourla, assured the public that ‘we will develop our product, develop our vaccine, using the highest ethical standards’. And the NHS has assured us of the same rigorous standards. So let’s take a look Pfizer’s history of ‘ethical standards’.
In 1992, Pfizer agreed to pay between $165 million and $215 million to settle lawsuits arising from the fracturing of the Bjork-Shiley Convexo-Concave heart valve, which by 2012 had resulted in 663 deaths.
Court approves settlement in Shiley heart-valve case Pfizer Inc. said Wednesday a federal judge has approved the previously announced agreement for settlement of claims to patients with the Bjork-Shiley… upi.com
In 1996, Pfizer conducted an unapproved clinical trial on 200 Nigerian children with its experimental anti-meningitis drug, Trovafloxacin, without parental consent, which led to the death of 11 children from kidney failure and left dozens more disabled. http://news.bbc.co.uk/1/hi/world/africa/6768799.stm …
In 2011, Pfizer paid just $700,000 to four families who had lost a child, and set up a $35 million fund for those disabled by their drug experiment. This cover-up was the basis to the John Le Carré book and film, The Constant Gardener.
Pfizer: Nigeria drug trial victims get compensation US-based pharmaceutical giant Pfizer makes the first compensation payment to Nigerian families affected by a controversial drug trial 15 year… bbc.co.uk
In 2004, Pfizer’s subsidiary, Warner-Lambert, was fined $430 million to resolve criminal charges and civil liabilities for the fraudulent promotion of its epilepsy drug, Neurontin, paying and bribing doctors to prescribe it for uses not approved by the FDA.https://www.justice.gov/archive/opa/pr/2004/May/04_civ_322.htm …
In 2009, Pfizer spent $25.8 million lobbying Congressional lawmakers and federal agencies like the Department of Health and Human Services. Image of government building columns Pfizer Inc Lobbying Profile Pfizer Inc spent $25,819,268 lobbying in 2009. See the details. opensecrets.org
Pfizer’s expenditure on federal lobbying between 2006 and 2014 came to $89.89 million. In 2019 the second largest pharmaceutical company in the world spent $11 million lobbying the US federal government. Image of government building columns Pfizer Inc Lobbying Profile Pfizer Inc spent $11,000,000 lobbying in 2019. See the details. opensecrets.org
In 2009, Pfizer paid the largest health care fraud settlement and criminal fine ever, paying $2.3 billion to avoid criminal and civil liability for fraudulently marketing its anti-inflammatory drug, Bextra, which the FDA had refused due to safety concerns.
Justice Department Announces Largest Health Care Fraud Settlement in American pharmaceutical giant Pfizer Inc. and its subsidiary Pharmacia & Upjohn Company Inc. (hereinafter together “Pfizer”) have agreed to pay $… justice.gov
In 2009, Pfizer paid $750 million to settle 35,000 claims that its diabetes drug, Rezulin, was responsible for 63 deaths and dozens of liver failures. In 1999, an epidemiologist at the FDA said Rezulin was ‘one of the most dangerous drugs on the market’. LA Times logo
Pfizer Agrees to Settle Suit Over Diabetes Drug Rezulin Pfizer Inc. agreed Friday to settle a lawsuit over the diabetes drug Rezulin after a jury earlier in the day awarded $43 million to a Texas woman wh… latimes.com
In 2010, Pfizer was ordered to pay $142.1 million in damages for violating a federal anti-racketeering law by its fraudulent sale and marketing of its epilepsy drug Neurontin for uses not approved by the FDA, including for migraines and bi-polar disorder.
Neurontin Lawsuits – Pfizer Illegal Marketing, Injury Claims Pfizer Marketing Illegal Neurontin. Pfizer Inc. has been ordered to pay $142.1M in damages for violating a federal antiracketeering law in its marketing. yourlawyer.com
In 2010, Pfizer admitted that, in the last 6 months of 2009 alone, it had paid $20 million to 4,500 doctors in the US for consulting and speaking on its behalf, and $15.3 million to 250 academic medical centres for clinical trials.
Pfizer admits paying $35 million to doctors over last 6 months Pfizer among other large pharmaceutical companies recently disclosed payments to doctors and other medical professionals for consulting and … news-medical.net
In 2012, Pfizer paid $45 million to settle charges of bribing doctors and other health-care professionals employed by foreign governments in order to win business. https://www.sec.gov/news/press-release/2012-2012-152htm …
The Chief of the Foreign Corrupt Practices Act Unit said: ‘Pfizer subsidiaries in several countries had bribery so entwined in their sales culture that they offered points and bonus programs to improperly reward foreign officials who proved to be their best customers’.
By 2012, Pfizer had paid $1.226 billion to settle claims by nearly 10,000 women that its hormone replacement therapy drug, Prempro, caused breast cancer.
In 2013, Pfizer agreed to pay $55 million to settle criminal charges of failing to warn patients and doctors about the risks of kidney disease, kidney injury, kidney failure and acute interstitial nephritis caused by its proton pump inhibitor, Protonix.
In 2013, Pfizer set aside $288 million to settle 2,700 claims that its stop-smoking drug, Chantix, caused suicidal thoughts and psychological disorders. The FDA subsequently determined that Chantix is probably associated with a higher risk of heart attack.
In 2013, Pfizer absolved itself of claims that its antidepressant, Effexor, caused congenital heart defects in the children of pregnant woman by arguing that the prescribing obstetrician was responsible for advising the patient about the medication’s use. https://europepmc.org/article/PMC/6424813 …
In 2014, Pfizer paid a further $325 million to settle a lawsuit brought by health-care benefit providers who claimed the company marketed its epilepsy drug, Neurontin, for purposes unapproved by the FDA.
In 2014, Pfizer paid $35 million to settle a law suit accusing its subsidiary of promoting the kidney transplant drug, Rapamune, for unapproved uses, including bribing doctors to prescribe it to patients.
In 2016, Pfizer was fined a record £84.2m for overcharging the NHS for its deregulated anti-epilepsy drug, Phenytoin, by 2,600% (from £2.83 to £67.50 a capsule), increasing the cost to UK taxpayers from £2 million in 2012 to about £50 million in 2013.
In May 2018, Pfizer had 6,000 lawsuits pending against claims that its testosterone replacement therapy products cause strokes, heart attacks, pulmonary embolism and deep vein thrombosis, and were marketed at healthy men for uses not approved by the FDA.
Pharmaceutical fraud – Wikipedia
$3 billion GSK settlement. On 2 July 2012, GlaxoSmithKline pleaded guilty to criminal charges and agreed to a $3 billion settlement of the largest health-care fraud case in the U.S. and the largest payment by a drug company. The settlement is related to the company’s illegal promotion of prescription drugs, its failure to report safety data, bribing doctors, and promoting medicines for uses for which they were not licensed. The drugs involved were Paxil, Wellbutrin, Advair, Lamictal, and Zofran for off-label, non-covered uses. Those and the drugs Imitrex, Lotronex, Flovent, and Valtrex were involved in the kickback scheme. The government investigation of GSK was launched largely on the basis of information provided by four whistleblowers who filed two qui tam (whistleblower) lawsuits against the company under the False Claims Act. GSK settled the whistleblowers’ lawsuits for a total of $1.017 billion out of the $3 billion settlement, the largest civil False Claims Act settlement to date.
Pfizer $2.3 billion settlement: Pfizer settled multiple civil and criminal allegations for $2.3 billion in the largest case of pharmaceutical and health care fraud in US history. The drugs involved were Bextra (an anti-inflammatory drug), Geodon (an anti-psychotic drug), Lipitor (a cholesterol drug), Norvasc (anti-hypertensive drug), Viagra (erectile dysfunction), Zithromax (antibiotic), Zyrtec (antihistamine), Zyvox (an antibiotic), Lyrica (an anti-epileptic drug), Relpax (anti-migraine drug), Celebrex (anti-inflammatory drug), and Depo-provera (birth control).
Merck $650 million settlement: Merck settled a nominal pricing fraud case in which the company was accused of taking kickbacks and violating Medicaid best price regulations for various drugs.
United States et al., ex rel. Jim Conrad and Constance Conrad v. Forest Pharmaceuticals, Inc, et al. involved a drug manufacturer selling a drug, Levothroid, that had never been approved by the FDA. These allegations settled for $42.5 million due to multiple whistleblowers stepping forward to provide detailed information on the alleged fraud. The collective reward to the relators in this case was over $14.6 million.
Wikipedia snapshot from 28 January 2021:
Article date: Monday 6 April 2009
At stake at one point last year was more than $8bn in punitive damages being sought in a string of cases, as well as potential jail terms in Nigeria for several Pfizer staff. “There has been a complex web of cases with proceedings in Connecticut, New York, Lagos, Abuja and Kano,” Mr Etigwe said. “The strategy of big companies when they are dealing with smaller opponents is to stretch the process, to overwhelm us until we are ready to accept whatever they want to offer.” Trovan never became the blockbuster that Pfizer had hoped for and it is no longer in production. The EU has banned the drug and it has been withdrawn from sale in the US.
- In July Health Secretary Matt Hancock claimed that conspiracy theorists are putting lives at risk
- The UK government’s Vaccine Damage Payment scheme is proof that vaccines can be unsafe
- Eligibility criteria Vaccine Damage Payment changed in 2015
- Update October 2020: AstraZeneca protected from vaccine liability
- Update November 2020: MHRA expects high volume of COVID-19 vaccine adverse drug reaction
- Update December 2020: Pfizer is given protection from legal action by the UK government
Discussion around vaccinations can be very contentious. There’s great nuance in this area and a short post will not do justice to the complex issues surrounding the usefulness and safety of vaccines. Nevertheless, while vaccines may have their role in protecting target populations from disease, not all have been proven safe to an acceptable level as shown in the resources below.
The UK government’s Vaccine Damage Payment scheme is probably the strongest proof that vaccines can be unsafe. Under the Vaccine Damage Payment scheme, people who have been severely disabled as a result of a vaccination against certain diseases can be eligible for a one-off tax-free payment of £120,000.
Conspiracy theorists are putting lives at risk?
It is an objective fact that a compensation scheme exists for those who have been damaged by vaccines. Nevertheless, Health Secretary Matt Hancock claimed that conspiracy theorists are putting lives at risk:
“Those who promulgate lies about dangers of vaccines that are safe and have been approved–they are threatening lives…”
Source: The Independent, 20 July 2020
Clearly, concerns about the safely of vaccines cannot be lies if there is a vaccine damage compensation scheme in place.
Eligibility changed in 2015
Eligibility requirements for vaccines covering certain diseases are listed and change over time. Interestingly, sometime around 2015, damage from vaccines for influenza caused by pandemics are explicitly listed as not eligible.
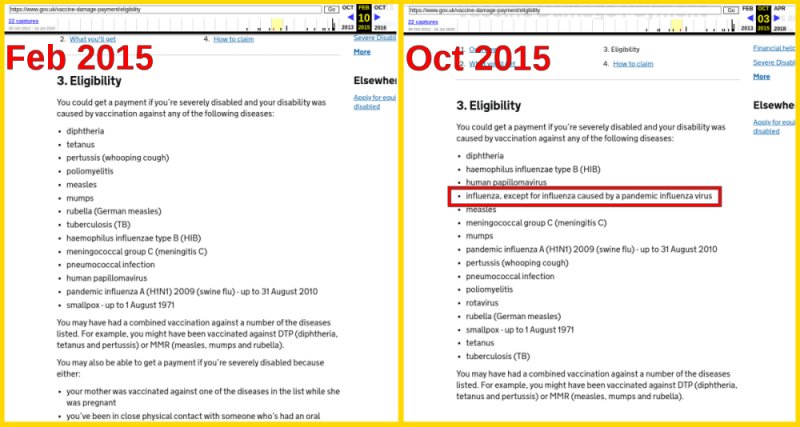
We do not know how the government compiles is eligibility criteria or why this change was made. However, it would be worthwhile to keep an eye on this list to see if the status of the upcoming COVID-19 vaccines.
AstraZeneca protected from vaccine liability
Update 1 August 2020: On 30 July 2020, Reuters reported that AstraZeneca, the UK government’s partner for developing its COVID-19 vaccine, will be exempt from coronavirus vaccine liability claims in most countries. The countries have not been named but Ruud Dobber, a member of Astra’s senior executive team, commented:
“This is a unique situation where we as a company simply cannot take the risk if in … four years the vaccine is showing side effects.
In the contracts we have in place, we are asking for indemnification. For most countries it is acceptable to take that risk on their shoulders because it is in their national interest.”
MHRA expects high volume of COVID-19 vaccine adverse drug reaction
Update November 2020: It came to light in mid-November that the UK’s Medicines & Healthcare products Regulatory Agency (MHRA) put out a contract award notice for an Artificial Intelligence (AI) software tool. It appears they expect a high volume of COVID-19 vaccine Adverse Drug Reaction (ADRs) from the upcoming vaccines:
…it is not possible to retrofit the MHRA’s legacy systems to handle the volume of ADRs that will be generated by a Covid-19 vaccine. Therefore, if the MHRA does not implement the AI tool, it will be unable to process these ADRs effectively.
Pfizer given legal indemnity
Update 2 December 2020: According to the Independent, Pfizer now has a legal indemnity from being sued by patients who develop any complications from its new mRNA vaccine that will be rolled out in the UK. NHS staff providing the vaccine will also be protected.
Resources
- UK Government Vaccine Damage Payment (gov.uk)
- Ministers lose fight to stop payouts over swine flu jab narcolepsy cases (The Guardian)
- Dengue vaccine fiasco leads to criminal charges for researcher in the Philippines (Science Magazine)
- Polio outbreaks in Africa caused by mutation of strain in vaccine (The Guardian)
- Pakistan accused of cover-up over fresh polio outbreak – (The Guardian)
- The Vaccination Debate (The Guardian)
- AstraZeneca to be exempt from coronavirus vaccine liability claims in most countries (Reuters)
- Zostavax Lawsuit (ClassAction.com)
- Pfizer to pay £50m after deaths of Nigerian children in drug trial experiment (The Independent)
- MHRA urgently seeks software tool to process the expected high volume of Covid-19 vaccine Adverse Drug Reaction (Tenders Electronic Daily)
- Pfizer given protection from legal action by UK government (The Independent)
View all articles related to COVID-19 and vaccination.